Chrome Yellow
Artificial inorganic pigmentComposition and Properties of Chrome Yellow
Chrome yellow is either lead chromate PbCrO4 or solid solutions of lead chromate and lead sulfate PbCrO4 · PbSO4.
All chromates are affected by acids and alkalis. Reaction with acids leads to the formation of orange dichromate ion Cr2O72-, in alkaline solutions the pale yellow chromate ion is formed. It is not lightfast and darkens with age. The degradation of this pigment has been extensively studied using x-ray fluorescence (2A – 2E). Lead chromate is compatible with all other pigments.
Video: 'Don't Fade Away - Saving the Vivid Yellows of Matisse and van Gogh' by Jennifer Mass
References
(1) Kühn, H. and Curran, M., Chrome yellow and Other Chromate Pigments, in Artists’ Pigments. A Handbook of Their History and Characteristics, Vol. 1: Feller, R.L. (Ed.) Oxford University Press 1986, pp. 187-218. Available as pdf from the National Gallery of Art.
(2 A) Monico L, Van der Snickt G, Janssens K, De Nolf W, Miliani C, Verbeeck J, Tian H, Tan H, Dik J, Radepont M, Cotte, M., Degradation process of lead chromate in paintings by Vincent van Gogh studied by means of synchrotron X-ray spectromicroscopy and related methods. 1. Artificially aged model samples, Anal Chem. 2011 Feb 15;83(4), 1214-23. doi: 10.1021/ac102424h.
(2 B) Same authors, Degradation process of lead chromate in paintings by Vincent van Gogh studied by means of synchrotron X-ray spectromicroscopy and related methods. 2. Original paint layer samples, Anal Chem. 2011 Feb 15;83(4), 1224-31. doi: 10.1021/ac1025122.
(2 C) Same authors, Degradation process of lead chromate in paintings by Vincent van Gogh studied by means of spectromicroscopic methods. 3. Synthesis, characterization, and detection of different crystal forms of the chrome yellow pigment. Anal Chem. 2013 Jan 15;85(2): 851-9. doi: 10.1021/ac302158b. Epub 2012 Dec 27.
(2 D) Same authors, Degradation process of lead chromate in paintings by Vincent van Gogh studied by means of spectromicroscopic methods. 4. Artificial aging of model samples of co-precipitates of lead chromate and lead sulfate, Anal Chem. 2013 Jan 15;85(2): 860-7. doi: 10.1021/ac3021592. Epub 2012 Dec 27.
(2 E) Same authors, Degradation process of lead chromate in paintings by Vincent van Gogh studied by means of spectromicroscopic methods. Part 5. Effects of nonoriginal surface coatings into the nature and distribution of chromium and sulfur species in chrome yellow paints, Anal Chem. 2014 Nov 4; 86(21): 10804-11. doi: 10.1021/ac502841g. Epub 2014 Oct 24.

Pigment

Painted swatch
Names of Chrome Yellow
Alternative names
Lemon yellow, king’s yellow, Leipzig yellow, Paris yellow, Vienna yellow, Cologne yellow
Color Index
PY 34, CI 77600, 77603
Word origin
After the element chromium, from Greek khrōma = color
Chromgelb
German
Jaune de chrôme
French
Giallo di cromo
Italian
Amarillo de cromo
Spanish
Preparation of Chrome Yellow
Attention: All chromates and dichromates are highly toxic chemicals and should not be used by people not trained to handle them.
Lead chromate can be prepared by mixing a solution of potassium chromate and lead (II)-nitrate. The resulting precipitate is then filtered, dried and homogenized in a mortar.
Video: 'Chromate Pigments' by The Alchemical Arts
References
(1) Ramette, R. W., Precipitation of lead chromate from homogeneous solution. A lecture demonstration or laboratory experiment, J. Chem. Educ., 49, 1972, s. 270.

Mineral crocoite from Tasmania
History of Use
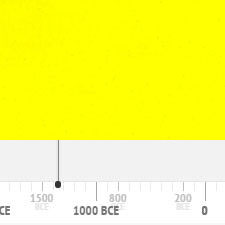
The French chemist Louis Vauquelin discovered the element chrome in 1797. Shortly after the possibility of using lead chromate as a pigment was recognized and the first methods of preparation were invented. Chrome yellow has been in use since the first quarter of the nineteenth century.
The following graph gives the frequency of its use in the paintings of the Schack Collection in the Bavarian State Art Collections in Munich (1).
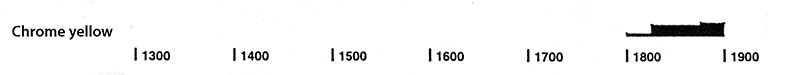
References
(1) Kühn, H., Die Pigmente in den Gemälden der Schack-Galerie, in: Bayerische Staatsgemäldesammlungen (Ed.) Schack-Galerie (Gemäldekataloge Bd. II), München 1969.
(2) Otero, V., Carlyle, L., Vilarigues, M., & Melo, M. J. (2012). Chrome yellow in nineteenth-century art: historic reconstructions of an artists’ pigment. RSC Advances, 2(5), 1798-1805. doi: 10.1039/C1RA00614B
(3) Marta Felix, Vanessa Otero, Joana Pinto, Marcia Vilarigues, Leslie Carlyle, Maria João Melo, Vanessa Matias, Barium, zinc and strontium yellows in late 19th–early 20th-century oil paintings, Heritage Science, 5:46, 2017. DOI 10.1186/s40494-017-0160-3
Identification
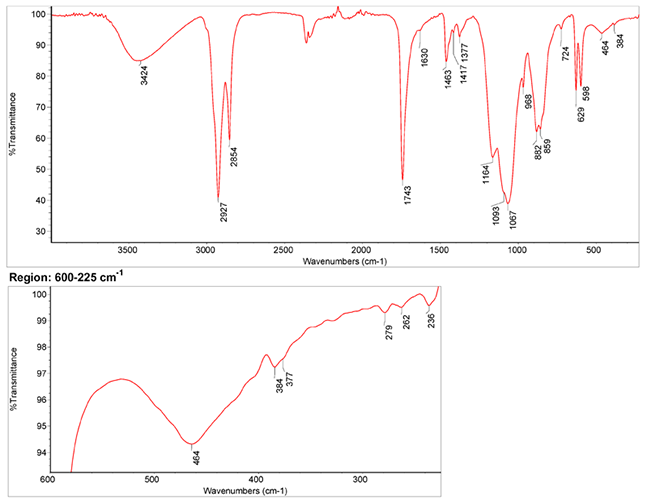
Infrared Spectrum
1. IR Spectrum of chrome yellow in the ATR-FT-IR spectra of different pure inorganic pigments, University of Tartu, Estonia.
2. IR Spectrum by NIST (National Institute of Standards and Technology)
3. IR Spectrum of chrome yellow in linseed oil by S. Vahur, Database of ATR-IR spectra of materials related to paints and coatings, University of Tartu, Estonia.
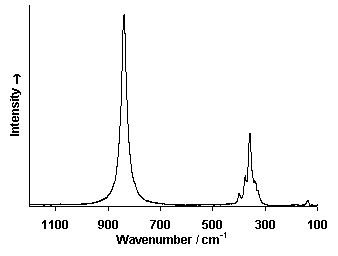
Raman Spectrum
Spectrum by Ian M. Bell, Robin J.H. Clark and Peter J. Gibbs, Raman Spectroscopic Library
University College of London
X-Ray Fluorescence Spektrum (XRF)
XRF Spectrum in the Free XRF Spectroscopy Database of Pigments Checker, CHSOS website.
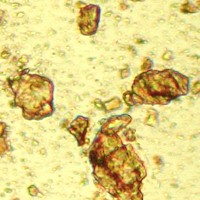
Microphotograph
image © Volker Emrath
Further Reading
References
(1) Kühn, H. and Curran, M., Chrome yellow and Other Chromate Pigments, in Artists’ Pigments. A Handbook of Their History and Characteristics, Vol. 1: Feller, R.L. (Ed.) Oxford University Press 1986, pp. 187-218. Available as pdf from the National Gallery of Art.
(2) S. Muntwyler, J. Lipscher, HP. Schneider, Das Farbenbuch, 2nd. Ed., 2023, alataverlag Elsau, pp. 102-103.