Cadmium Yellow
Artificial inorganic pigmentComposition and Properties of Cadmium Yellow
Cadmium yellow is cadmium sulfide CdS. There are many different color variations achieved by an admixture of zinc sulfide (light yellow), and selenium sulfide (orange to red). The variations are solid solutions of either zinc sulfide or selenium sulfide in cadmium sulfide.
It is stable as a pure pigment. The occurrences of bleaching or darkening in the beginnings of its use were due to impurities (1). Changes in the pigment with time were investigated by several methods such as x-ray fluorescence and infrared spectroscopy (2,3). The pigment is lightfast and there are no reports to the contrary, but it is not compatible with lead and copper-containing pigments as they form dark sulfides of lead or copper.
Video: 'Don't Fade Away - Saving the Vivid Yellows of Matisse and van Gogh' by Jennifer Mass
References
(1) Fiedler, I., Bayard, M.A., Cadmium Yellows, Oranges, and Reds, in Artists’ Pigments. A Handbook of Their History and Characteristics, Vol. 1: Feller, R.L. (Ed.) Oxford University Press 1986, p. 65 – 108. Available as pdf from the National Gallery of Art.
(2) E. Pouyet, M. Cotte, B. Fayard, M. Salomé, F. Meirer, A. Mehta, E. S. Uffelman, A. Hull, F. Vanmeert and 5 more, 2D X-ray and FTIR micro-analysis of the degradation of cadmium yellow pigment in paintings of Henri Matisse, Applied Physics A, November 2015, Volume 121, Issue 3, pp 967-980.
(3) Geert Van der Snickt, Koen Janssens, Joris Dik, Wout De Nolf, Frederik Vanmeert, Jacub Jaroszewicz, Marine Cotte, Gerald Falkenberg, Luuk Van der Loeff, Combined use of Synchrotron Radiation Based Micro-X-ray Fluorescence, Micro-X-ray Diffraction, Micro-X-ray Absorption Near-Edge, and Micro-Fourier Transform Infrared Spectroscopies for Revealing an Alternative Degradation Pathway of the Pigment Cadmium Yellow in a Painting by Van Gogh, Analytical Chemistry 08/2012; 84(23). DOI:10.1021/ac3015627 · 5.64.
Names of Cadmium Yellow
Alternative names
Aurora yellow, jaune brilliant
Color Index
PY 37, CI 77199
Word origin
Word coined in Modern Latin from cadmia, a word used by ancient naturalists for various earths and oxides (especially zinc carbonate), from Greek kadmeia “Cadmean (earth),” from Kadmos “Cadmus,” legendary founder of Boeotian Thebes. So called because the earth was first found in the vicinity of Thebes (Kadmeioi was an alternative name for “Thebans” since the time of Homer).
From WordFinder
Cadmiumgelb
German
Jaune de cadmium
French
Giallo di cadmio
Italian
Amarillo de cadmio
Spanish
Preparation of Cadmium Yellow
Attention: Cadmium compounds are highly toxic and should not be handled by people not trained to do so.
Cadmium yellow can either be prepared by heating of metallic cadmium with sulfur or by precipitation of the unsoluble cadmium sulfide by a reaction of a solution of a cadmium salt, such as cadmium chloride, with a solution of sodium sulfide. The pigment can also be prepared in the laboratory provided it is equipped with a good functioning fume hood as the gaseous hydrogen sulfide H2S which is produced when sodium sulfide is solved in water is poisonous and has a very unpleasant odor (the same as rotten eggs).
Preparation of the Pigment in the Lab
A solution of 9,7 g cadmium nitrate Cd(NO3)2 · 4 H2O in 50 ml deionized water is poured into the solution of 8,3 g sodium sulfide Na2S · 9 H2O in 50 ml deionized water while mixing. The resulting precipitate is then filtered, dried and homogenized in a mortar.



Video: 'Preparation of the Pigment' by The Alchemical Arts
Cadmium sulfide occurs naturally as the mineral greenockite but the mineral has never been used as a pigment.
Mineral Greenockite

image Ch. Rewitzer
History of Use
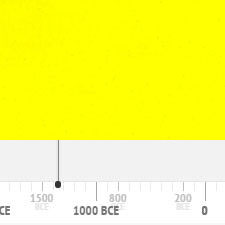
Cadmium pigments have been in use since about 1840 until today. They might get banned in the European Union in the next future because of their toxicity.
Examples of use
Claude Monet, Water-Lilies, after 1916

2 Bright yellow impasto (paint laid on an area very thickly): cadmium yellow.

Identification
Fiber Optics Reflectance Spectra (FORS)
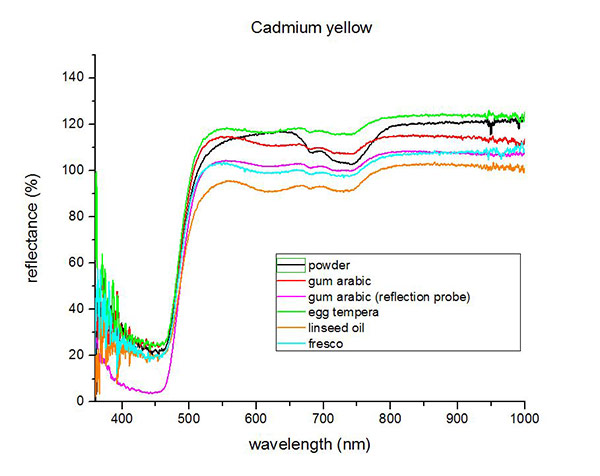 Spectra by A. Cosentino, Cultural Heritage Science Open Source (CHSOS)
Spectra by A. Cosentino, Cultural Heritage Science Open Source (CHSOS)
Infrared Spectrum
1. IR Spectrum of cadmium yellow in the ATR-FT-IR spectra of different pure inorganic pigments, University of Tartu, Estonia. 2. IR Spectrum of cadmium yellow in linseed oil by S. Vahur, Database of ATR-IR spectra of materials related to paints and coatings, University of Tartu, Estonia
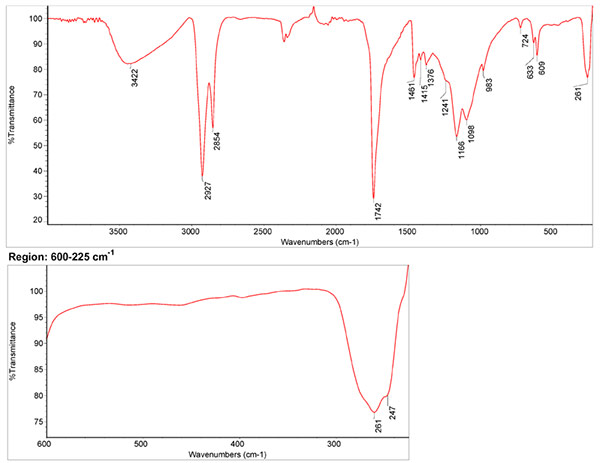 Spectrum by S. Vahur, Database of ATR-IR spectra of materials related to paints and coatings, University of Tartu, Estonia
Spectrum by S. Vahur, Database of ATR-IR spectra of materials related to paints and coatings, University of Tartu, Estonia
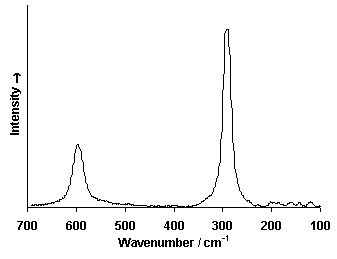
Raman Spectrum
Spectrum by Ian M. Bell, Robin J.H. Clark and Peter J. Gibbs, Raman Spectroscopic Library University College of London
X-Ray Fluorescence Spektrum (XRF)
XRF Spectrum in the Free XRF Spectroscopy Database of Pigments Checker, CHSOS website.
References
(1) Thoury M, Delaney JK, Rie ER, Palmer M, Morales K, Krueger J., Near-infrared luminescence of cadmium pigments: in situ identification and mapping in paintings, Appl Spectrosc. 2011 Aug;65(8):939-51. doi: 10.1366/11-06230.
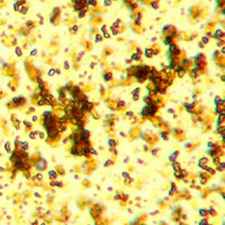
Microphotograph
image © Volker Emrath
Further Reading
References
(1) Fiedler, I., Bayard, M.A., Cadmium Yellows, Oranges, and Reds, in Artists’ Pigments. A Handbook of Their History and Characteristics, Vol. 1: Feller, R.L. (Ed.) Oxford University Press 1986, p. 65 – 108. Available as pdf from the National Gallery of Art.
(2) S. Muntwyler, J. Lipscher, HP. Schneider, Das Farbenbuch, 2nd. Ed., 2023, alataverlag Elsau, pp. 106-107.